Abstract
Introduction: Platelet glycoproteins are key contributors to platelet function but their glycans structure is unclear. Alterations in glycan composition have been reported to impact platelet clearance under physiological conditions and in the disease mechanism of immune thrombocytopenia (ITP). Therefore, this study sought to characterize glycan structures in human platelets from healthy control individuals and ITP patients using mass spectrometry (MS)-based glycomics approach, andto compare their glycomic profiles to facilitate understanding of glycan alterations in ITP.
Methods: Glycan residues on platelet surface were determined by flow cytometry. Platelet lysates (1×10 8 platelets) from 4 healthy controls and from 4 ITP patients with a clear anomalous glycosylation pattern were characterized by MALDI-MS based glycomic approaches. N-linked glycans were released from platelet glycoproteins by PNGase F digestion and subsequently purified with a Sep-Pak C18 reverse phase cartridge. O-linked glycans were released by reductive elimination. Both pools of glycans were permethylated prior to MALDI-TOF MS to obtain an initial carbohydrate profile. Selected glycan molecular ion species were analyzed by MALDI-TOF-TOF MS/MS before and after digesting with exoglycosidases.
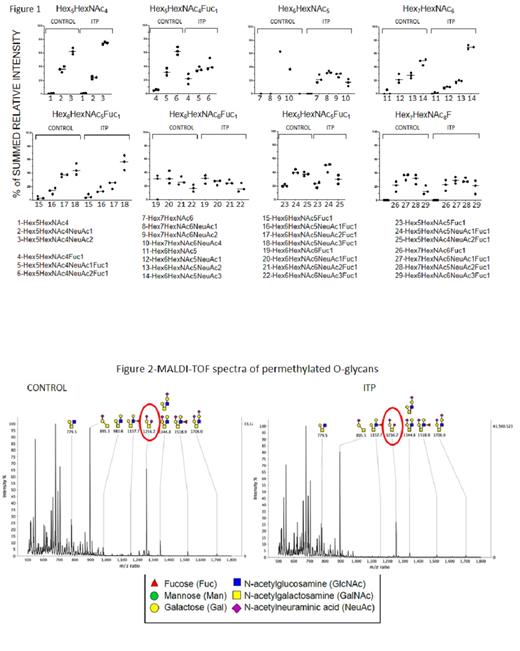
Results: Glycans present in platelets from healthy controls and from ITP patients were largely consistent. The MS spectra for N-glycans showed a mixture of high mannose glycans (m/z 1579.8, 1783.9, 1988.0, 2192.1 and 2396.2); complex glycans (m/z 2966.5, 3776.9 and 4587.4); and bisected or truncated glycans (m/z 3211.6 and 4022.1). The spectra showed the presence of bi-, tri- and tetra-antennary complex glycans, which varied in their level of sialylation.Figure 1 shows the relative abundance ratio within eight families of core glycan structures. Platelets from ITP patients showed a consistent increase in desialylated structures such as Hex 5HexNAc 4Fuc 1.
In addition to different amounts of attached sialic acid, varying levels of fucosylation were observed; ranging from the addition of a single core Fuc (m/z 2244.1), to the addition of up to three Fuc residues (m/z 3402.7).
Collision-activated decomposition (CAD) MALDI-TOF/TOF analysis was performed to generate fragment ions from molecular ions detected in MALDI-TOF profiling for detailed sequencing of platelet N-glycans. This analysis suggested the presence of three isoforms: (i) sialylated tetra-antennary structure with core fucosylation and one Lewis x/a antenna; (ii) sialylated tetra-antennary structure with one Lewis y/b antenna; (ii) sialylated, core-fucosylated tri-antennary structure with one LacNAc extension and one Lewis x/a antenna.
Sialidase S digestion was used to highlight the extent of desialylation in the presence of sialidase. Noteworthy, the ratio of non-sialylated bi-and tri-antennary N-glycans in ITP patients were higher than that in controls. This enzymatic digestion confirms the presence of α2,3-linked Neu5Ac on platelet glycans. Remaining sialylated structures may possess α2,6-linked Neu5Ac.
O-glycan profiles obtained showed the predominance of core 1 and core 2 structures (Figure 2). The two most dominant glycan structures in core 1 were sialyl T antigen (GalNAc 1Gal 1NeuAc 1; m/z 895.5) and disialyl T antigen (GalNAc 1Gal 1NeuAc 2; m/z 1256.7), being the latter less abundant in ITP patients. The core 2 structure was modified by the addition of fucose and/or sialic acid residues (m/z 1157.7, 1344.8, 1518.9 and 1706.0).
Conclusion: N- and O-glycan structures in human platelets were characterized by MALDI-TOF MS profiling to reveal interesting structural features including the presence of sialylLewis x/a epitope, Lewis x/a epitope, Lewis y/b epitope and LacNAc extensions in complex type N-glycans. Presence of terminal sialic acid and sialylLewis x/a on platelet N-glycan antenna also suggest their potentialfunction as ligands for siglecs that are associated with cell signaling functions. Siglec-1 and -2 have been suggested to have potential roles in ethiopathogenesis of autoimmune diseases. Desialylation observed in glycans of platelets from ITP patients, might trigger immune system activation in these patients.
This research was funded by ISCIII-Fondos FEDER PI19/00772 and Platelet Disorder Support Association
Butta: Roche: Speakers Bureau; Takeda: Research Funding, Speakers Bureau; CSL-Behring: Research Funding; Novo-Nordisk: Speakers Bureau. Canales: F. Hoffmann-La Roche Ltd: Consultancy, Honoraria, Speakers Bureau; Karyopharm: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Speakers Bureau; iQone: Honoraria; Sandoz: Honoraria, Speakers Bureau; Incyte: Consultancy; Janssen: Consultancy, Honoraria, Speakers Bureau; Gilead/Kite: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Sanofi: Consultancy; Eusa Pharma: Consultancy, Honoraria; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria. Jiménez-Yuste: Pfizer: Consultancy, Honoraria, Research Funding; F. Hoffmann-La Roche Ltd: Consultancy, Honoraria, Research Funding; BioMarin: Consultancy; Sobi: Consultancy, Honoraria, Research Funding; NovoNordisk: Consultancy, Honoraria, Research Funding; Octapharma: Consultancy, Honoraria, Research Funding; Sanofi: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Research Funding; Bayer: Consultancy, Honoraria, Research Funding; CSL Behring: Consultancy, Honoraria, Research Funding; Grifols: Consultancy, Honoraria, Research Funding. Alvarez Román: Octapharma: Consultancy, Honoraria, Research Funding; Biomarin: Consultancy, Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Bayer: Consultancy, Honoraria, Research Funding; CSL-Behring: Consultancy, Honoraria, Research Funding; Grifols: Consultancy, Honoraria, Research Funding; Novo-Nordisk: Consultancy, Honoraria, Research Funding; Sobi: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal